Zymo Dna Rna Extraction Protocol
ZYMO Quick-DNA/RNA Miniprep Plus Kit Troubleshooting
Goal
- To determine if both DNA and RNA extracts can be obtained from an adult coral homogenate (see airbrushing protocol) and a biopsy of adult tissue and skeleton. The coral used for this test was Porites astreoides collected from Bermuda.
Experimental design
Table 1: Experimental Design to test DNA/RNA extraction methods on 2 types of coral tissues.
| Sample # | Sample Type | Coral ID/Vial# | Volume/Size of Sample |
|---|---|---|---|
| 1 | Homogenate | 951 | 500 mL |
| 2 | Homogenate | 954 | 500 mL |
| 3 | Fragment | R20-A | ~10 mm diameter |
| 4 | Fragment | R20-A | ~10 mm diameter |
Overall workflow
- Prepare samples
- Extract and prep DNA with column (elute RNA and Protein)
- Extract and prep RNA with column (elute Protein)
- Quantify DNA, RNA, and protein extracts
Protocol preparation
- Using slightly modified Zymo Duet DNA/RNA extraction protocolwhich will extract both DNA and RNA at the same time (Below are summary steps)
Reagents and supplies
- RNase-free Water
- 100% ethanol, ACS grade or better
- 10mM Tris HCl pH 8.0 made with RNase-free water
- ZR BBashing lysis tubes with 0.1mm and 0.5mm beads
Equipment
- Rocking oven that can be set to 55°C
- RNase away and a designated RNase free space
- Tabletop and larger centrifuges for 1.5mL and 50mL tubes capable of 12,000 x g
Clipper Sterilization
- Rub down clippers with:
- 10% Bleach solution
- DI water
- 90% ethanol
- RNAse free water
Notes before starting
- Wipe down benchtop with RNase away and have the spray bottle and kimwipes on-hand to use frequently
Sample preparation
Homogenate preparation
- Immediately after taking sample out of the -80°C freezer, add 500μL of DNA/RNA shield
- Add 100 μL of Protenase K digestion buffer
- Derived from volume of homogenate and 300/30 ratio from Zymo Protocol
- Add 50 μL of Protenase K
- Derived from volume of homogenate and 300/15 ratio from Zymo Protocol
- Incubate on Thermomixer at 56°C at 600 rpm for 30 minutes
- Remove all liquid (1150 μL) and transfer in 5 mL tubes
Fragment preparation
- Sterilize clippers (as outlined above)
- Clip off ~10mm in diameter of tissue and skeleton and place into ZR BBashing lysis tube
- Add 1000 μL of DNA/RNA shield (may be too much)
- Vortex for ~1 minute
- Remove 500 μL of supernatant and transfer into 5mL tube
- Large mucus chunks made this step difficult –> potential source of error
DNA Extraction
- Add equal volumes of DNA lysis buffer as sample volume to the 5 mL tube with sample
- 1150 μL for the homogenate samples
- 500 μL for the fragment samples
- Vortex to mix
- Transfer 700 μL into DNA filter column (yellow)
- Centrifuge at 16,000 rcf for 30 seconds
- Remove flow through liquid and transfer into a new 5 mL tube labeled for RNA
- Repeat steps 2 until all liquid is gone
- Add 400 μL of DNA/RNA prep buffer to column
- Centrifuge at 16,000 rcf for 30 seconds
- Remove the flow through and transfer to waste
- Add 700 μL of DNA/RNA wash buffer
- Centrifuge at 16,000 rcf for 30 seconds
- Remove the flow through and transfer to waste
- Add 400 μL of DNA/RNA wash buffer
- Centrifuge at 16,000 rcf for 2 minutes
- Remove the flow through and transfer to waste
- Removed column and place into a new sterile 1.5 mL tube
- Add 50 μL of 10 mM Tris HCL (warmed to 55°C) directly to filter
- Incubate at room temperature for 5 minutes
- Centrifuge at 16,000 rcf for 30 seconds
- Keep flow through in tube
- Add another 50 μL of 10 mM Tris HCL (warmed to 55°C) directly to filter
- Incubate at room temperature for 5 minutes
- Centrifuge at 16,000 rcf for 30 seconds
- Discard column and keep flow through in tube
- Aliquot into appropriate tubes for storage at -20°C
RNA Extraction
- Add equal volumes of 100% Ethanol as sample volume to the 5 mL tube with sample
- 1150 μL for the homogenate samples
- 500 μL for the fragment samples
- Vortex to mix
- Transfer 700 μL into RNA filter column (green)
- Centrifuge at 16,000 rcf for 30 seconds
- Remove flow through liquid and transfer into a new 5 mL tube labeled for Protein
- Repeat steps 12 until all liquid is gone
- Add 400 μL of DNA/RNA wash buffer to column
- Centrifuge at 16,000 rcf for 30 seconds
- Remove the flow through and transfer to waste
- Make DNase I master mix: [75μL DNA digestion buffer and 5μL DNase] X n (sample #)
- Add 80μL of DNase I master mix directly to the filter of each column. Incubate at room temperature for ~15 minutes
- Add 400 μL of DNA/RNA prep buffer to column
- Centrifuge at 16,000 rcf for 30 seconds
- Remove the flow through and transfer to waste
- Add 700 μL of DNA/RNA wash buffer
- Centrifuge at 16,000 rcf for 30 seconds
- Remove the flow through and transfer to waste
- Add 400 μL of DNA/RNA wash buffer
- Centrifuge at 16,000 rcf for 2 minutes
- Remove the flow through and transfer to waste
- Removed column and place into a new sterile 1.5 mL tube
- Add 50 μL of RNAase-free water (warmed to 55°C) directly to filter
- Incubate at room temperature for 5 minutes
- Centrifuge at 16,000 rcf for 30 seconds
- Keep flow through in tube
- Add another 50 μL of RNAase-free water (warmed to 55°C) directly to filter
- Incubate at room temperature for 5 minutes
- Centrifuge at 16,000 rcf for 30 seconds
- Discard column and keep flow through in tube
- Aliquot into appropriate tubes for storage at -80°C
Quantification
Qubit
- For the Qubit readings, 10 μL of each standard were used and 1 μL of each sample were used.
Table 2: Qubit readings for the extracted DNA and RNA samples.
| Standard 1 | Standard 2 | #1 (ng/uL) | #2 (ng/uL) | #3 (ng/uL) | #4 (ng/uL) | |
|---|---|---|---|---|---|---|
| DNA | 205.05 | 22675.65 | 16.70 | 13.00 | 8.24 | 3.03 |
| DNA | NA | NA | 16.10 | 13.00 | 8.10 | 2.98 |
| RNA | 393.14 | 11692.94 | 10.20 | 11.80 | 12.60 | Too low |
| RNA | NA | NA | 10.00 | 11.60 | 12.40 | Too low |
Tapestation
- Only RNA samples were run on the tape station
- Due to the low sample reading from sample #4, it was not run on the tapestation
Tapestation results
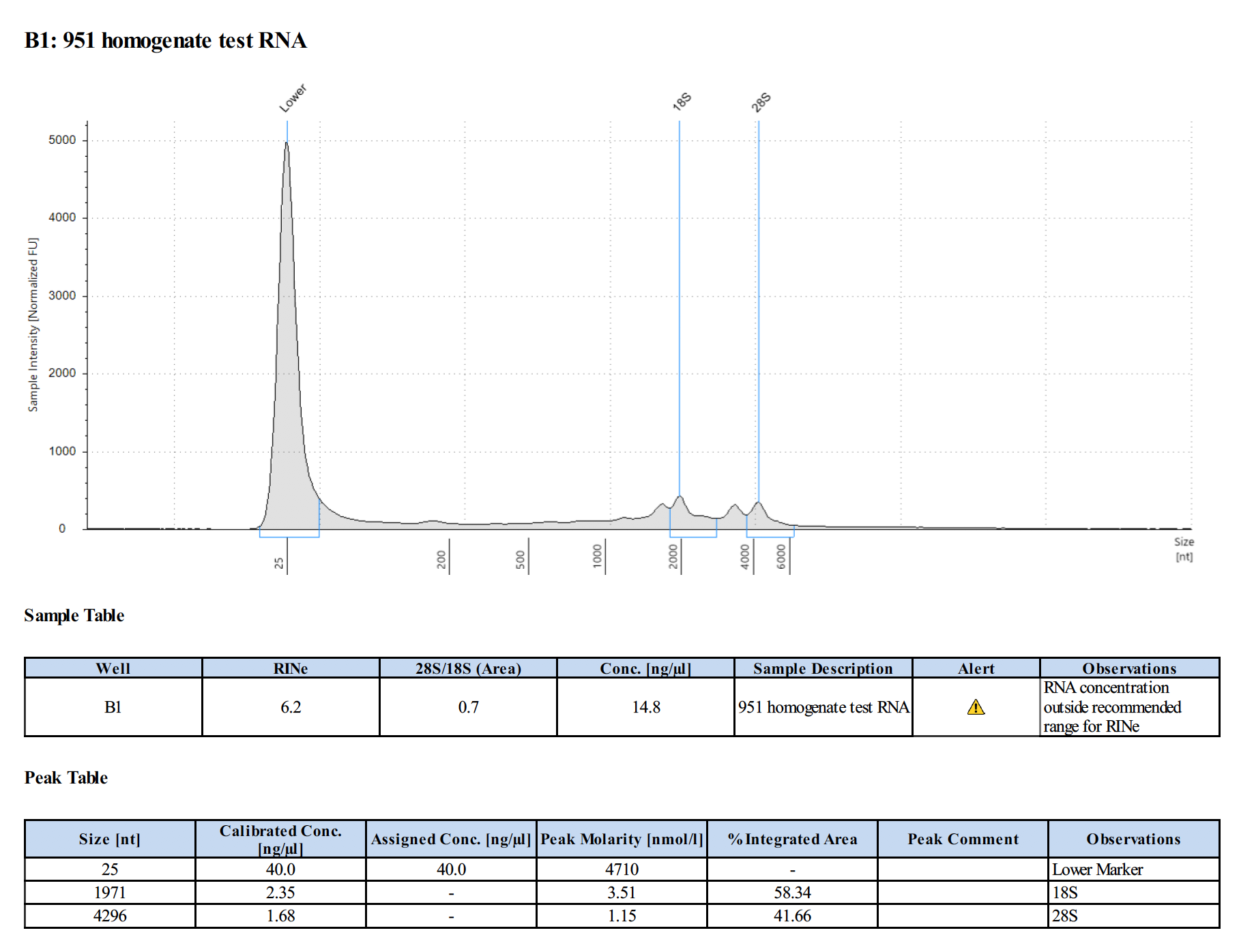
Sample 1:
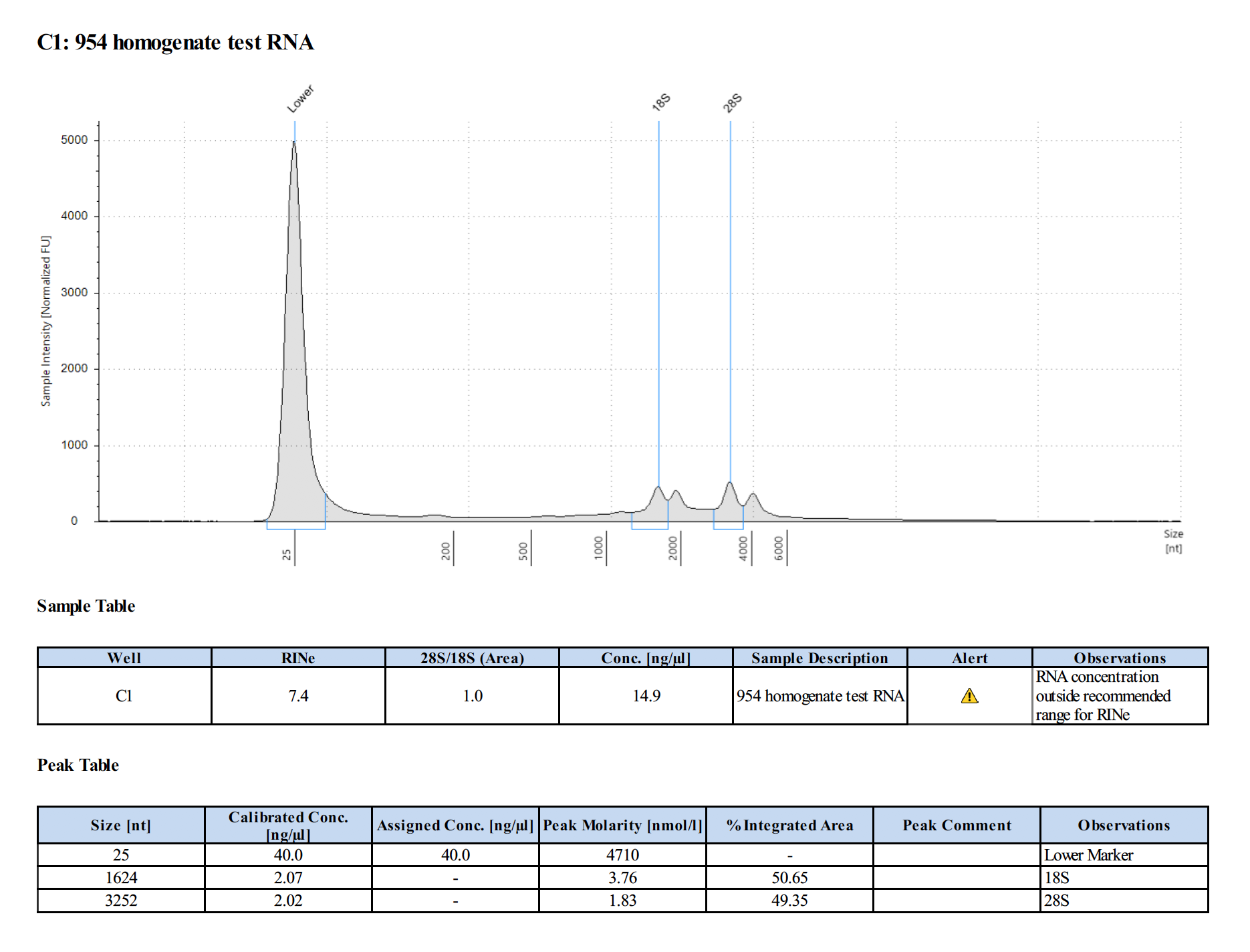
Sample 2:
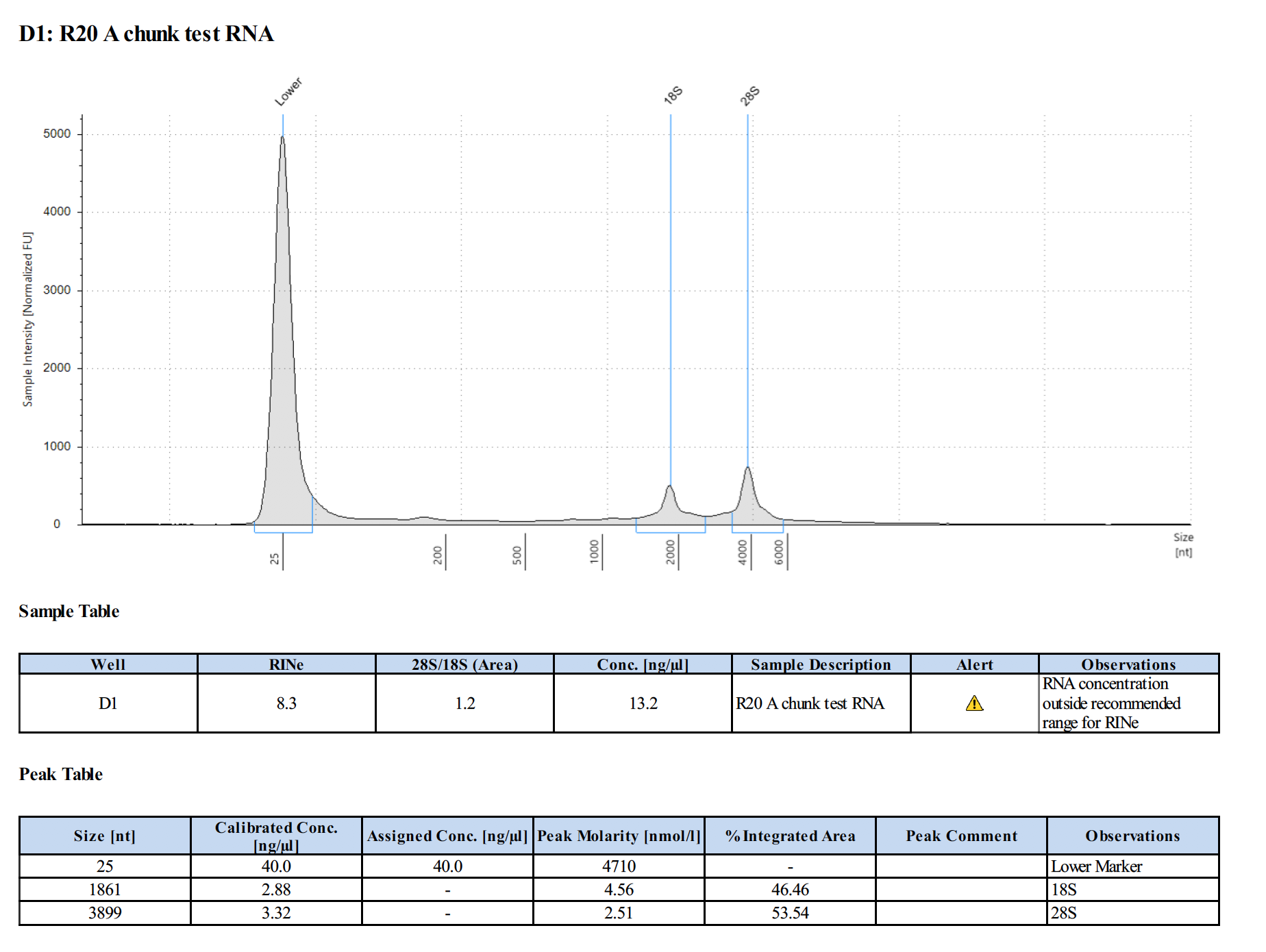
Sample 3:
Conclusions
- Homogenates from airbrushed coral tissue can only be used for DNA, RNA is too degraded
- Need to troubleshoot homogenizing coral fragment with beadbeating, but it is possible to extract RNA using this protocol
Written on February 13, 2019
